Brewing is not always a ‘walk in the park’. In fact, the art of brewing is not just in juggling flavors and recipes, it also requires a thorough scientific knowledge to manage the brewing process.
It would be a shame, wouldn’t it, if a mistake in brewing caused a fantastic beer recipe to go down the drain because of an off-flavour?
Overview
- Diacetyl: Butter, buttermilk, popcorn
- DMS (dimethyl sulfide): Cooked corn, broccoli, vegetable oil
- Skunk
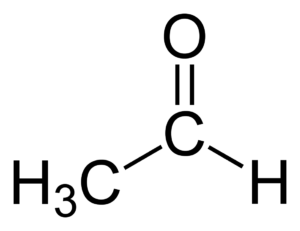
- Acetaldehyde: Green apple, cucumber, fresh cut grass
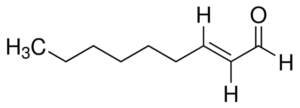
- Oxidation: Wet, sticky paper or cardboard
- Autolysis: Yeast, meat, barbecue chips, sulfur
- Gushing: Dust, old and damp cloth


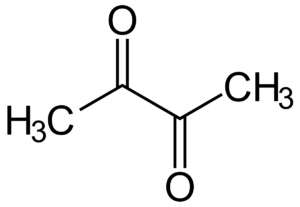
1. DIACETYL: butter flavor
If your beer tastes a little ‘greasier’ than usual, this may be due to diacetyl. This substance is sometimes allowed in low intensity in British beers such as Scottish ales and some lagers. In most cases, however, this is undesirable.
In its original form, α-acetolactate (AAL), the substance is tasteless and odorless. However, AAL spontaneously converts to diacetyl, which can negatively affect flavor.
“How do I prevent diacetyl?”
1) Allow to ferment long enough
During fermentation, yeast normally causes diacetyl to be converted into alcohol. Therefore, it is important that the beer has enough time to ferment. With top fermentation, diacetyl remains present for about 2 weeks, with bottom fermentation it is about 4 weeks.
2) Beware of mutated yeast
Due to a defect, the yeast may not be able to convert the diacetyl. Thus, diacetyl remains in the beer.
3) Keeping pipes clean
Last but not least, bacteria (lactobacillus, peciococcus,…) can also create diacetyl. This is a risk that both the brewer and the bartender can run since it is almost always caused by poor hygiene in the pipes. Enough rinsing and cleaning of the pipes can prevent this!
How do I spot diacetyl?
The taste and smell of diacetyl in beer is often described as a kind of
buttery -, popcorn flavor. No coincidence since it is a substance used in making popcorn.
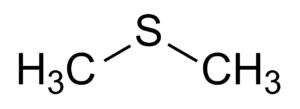
2. DMS or DIMETHYLSULFIDE: cooked vegetables
DMS, or dimethyl sulfide in full, is a substance formed during the boiling of beer. In its original form, it is still S-Methyl Methionine (SMM); an amino acid formed during malting. SMM is less common in two-row malt (two rows of grains) than in six-row malt (six rows of grains).
Since most beers contain a small amount of pale malt, there is a little SMM in almost every beer. So DMS is often present in beer to a slight degree, this is desirable in some lagers for example.
“How do I avoid DMS?”
1) Beware of mistakes in the cooking process
DMS is a highly volatile substance, which evaporates along with the water. So it is important that our brew bubbles well and boils long enough.
A common mistake is to cover the boil or not ventilate enough. Thus, the evaporated DMS will stick to the lid and eventually end up back in the beer.
2) Watch out for infections
Something that occurs less frequently but is still important to know is that an infection can also be the cause of DMS. In this case, however, the substance will impart a different taste than in the faulty boil.
3) Cool fast enough after cooking
SMM is converted into DMS by the heat. So if you let your beer cool down too slowly after boiling, it may happen that the remaining SMM remnants are still converted into DMS.
It is generally said that for every hour that your wort is still warm, 30% DMS is added.
How do I spot dimethyl sulfide?
The taste of this brew is often described as that of cooked vegetables, since when you cook vegetables you naturally get DMS as well. More specifically, the taste is very similar to that of canned corn.
DMS formed by infection tastes slightly different and is compared to the smell of lettuce.
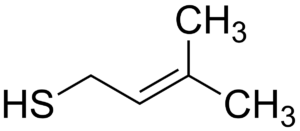
3. SKUNK; the aroma you definitely want to avoid.
Hops are one of the most important ingredients for brewing beer. You can hardly find beers without hops. It is therefore a fantastic ingredient that gives both flavor, aroma and bitterness to your beer.
Hops do have a major archenemy, namely UV light.
The interaction between these two causes the iso-alpha acids (IAA) in hops to be converted to 3-methyl-2-butene-1-tiol or in human language; skunk. The formation of skunk should be resisted at all times, as it creates an extremely unpleasant smell and taste.
“How do I prevent skunk?”
1) Avoid sunlight or harmful lighting
The first tip is obvious but also the most important of all. Sunlight contains UV light so one must pay attention to where you store beer from the beginning of the brewing process to the consumer or merchant. Be aware that some lamps also contain UV light!
Skunk can be formed in seconds so take plenty of precautions and provide storage areas with light rays without UV. Traders and consumers themselves should also take responsibility and store the beer in dark rooms.
2) Take a brown bottle or can
The color of the beer bottle can prevent many problems. It is no accident that almost every beer is packaged in brown bottles. These bottles block 98% of harmful light so shielding is still necessary here too!
Green bottles block about 20% and the ordinary ready-made bottles let all the harmful light through. A can is the ideal packaging against UV light, because it lets 0% harmful light through and then you are sure that no skunk will be formed. However, this is often too expensive a solution.
3) THIAA
After research by some of the beer giants, a solution to skunk called tetrahydro-iso-alpha-acids, or THIAA for short, has emerged.
THIAA is 100% stable and makes it impossible to form skunk. However, only bitter hops can then be used in the beer, making it not suitable for every beer.
How do you spot skunk?
It is not very difficult to spot skunk in your beer, as it has a very sharp, unpleasant smell. In nature this occurs in the skunks, their glands produce the smell skunk and they use this as a defense mechanism against enemies.
Not familiar with the smell of a skunk? Then let it be known that if you have a very unpleasant smell, don’t look far for the cause; this is probably skunk.
Some people are so sensitive to this that they can’t have a glass of beer in the sun.
4. ACETALDEHYDE; green apple
Acetaldehyde is created when glucose is converted to ethanol. It is therefore something that occurs during primary fermentation. At a later stage, the yeast will convert the acetaldehyde back into alcohol, the remaining value is often too small to observe.
If the residual value does appear remarkably high, the cause is often problematic fermentation or alcohol oxidation.
“How do I prevent acetaldehyde?”
1) Pay enough attention to fermentation
Yeast is the most important thing to avoid acetaldehyde. So, by all means, start by pitching enough yeast (adding yeast to the wort to initiate the initial fermentation process). Next, make sure the yeast has enough oxygen to multiply.
Don’t rush it, let the beer ferment completely and avoid oxygen as much as possible after it is finished.
2) Ensure good hygiene
Like many problems, acetaldehyde can be avoided by keeping everything clean and cleaning enough.
3) Add yeast if necessary
It can always happen that you do end up with acetaldehyde by, for example, taking the beer off fermentation too quickly. One can solve this by adding new yeast or using krausening (using fermented wort to ferment your beer in the bottle).
How do I spot acetaldehyde?
Acetaldehyde is actually the substance that is made in your body from alcohol. So it plays a big role in hangovers! Logically, it is harmful to a great extent and also tastes unpleasant.
The flavor of this brew is similar to the sour taste of green apples. The aroma tends more toward paint.
5. OXIDATION; wet cardboard
Oxidation of beer can happen at different times. The flavor defect is usually produced during the breakdown of unsaturated fatty acids and is caused during aldol condensation (the organic formation of aldehyde + alcohol).
The substance produced is trans-2-nonenal.
It provides a specific aroma and flavor that aged beers often exhibit. Since the aging process is almost always the cause of unwanted oxidation, the responsibility lies primarily with the merchants/consumers rather than the brewers themselves.
“How do I prevent oxidation?”
1) Inform customers as best as possible
Sometimes traders or end consumers don’t know what things to look out for to avoid oxidation. So sell beers to traders with a certain order, FIFO (First In First Out) is always the best idea. Stating an appropriate expiration date is mandatory in Belgium anyway, make it as accurate as possible.
Temperature plays a major role in storage. In a warm environment, beer matures more quickly and therefore causes unwanted oxidation. It is therefore always advisable to find a cool place to store the beer so that the taste remains as it should. Temperature fluctuations are also not done. So our tip is: communicate this to the customers.
2) Ensure appropriate distribution
When distributing, be sure to try to ensure that the transport is as cool as possible. Additionally, it is advisable to ask the transporter not to shake the beer too much, as swirling beer in the bottle can accelerate oxidation.
3) Watch out for oxygen in the bottle
It is important to make sure that there is not too much oxygen in the bottle, as with low filling. It is even recommended that before filling the bottle, you fill it with CO2 gas. That way you push the O2 out.
4) The more hops the better
The hoppier the beers are, the longer it takes for oxidation to occur. Hops inhibit the oxidation of beer, which is also the reason why hops were added back in the day: to preserve beer longer.
How do I spot oxidation?
As a brewer, it is often quite easy to taste oxidation. If the beer tastes different than it should after some storage time, oxidation is often the cause. The taste and aroma is often compared to wet paper or cardboard. Anyone who has experience with beer that has been stored too warm will recognize the taste.
6. AUTOLYSE; exaggerated yeast taste
Autolysis, literally translated as “self-destruction,” means the disintegration of dead yeast cells.
This releases the contents of the yeast cells. A large part of these contents consists of amino acids and enzymes. If these end up in the beer, they influence the taste, among other things.
“How do I prevent autolysis?”
1) Choose quality yeast
Weak, unhealthy yeast cells will self-destruct much faster than strong yeast cells. It is therefore obvious that quality yeast reduces the chance of autolysis quite a bit.
2) Watch your temperature during fermentation
Autolysis can be caused by heating or cooling the beer too quickly during fermentation.
Too high a temperature during and after fermentation also increases the risk of autolysis.
3) Ensure adequate oxygen
If the yeast cells cannot take in enough oxygen while multiplying, they will become weaker and start destroying themselves more quickly.
So good wort aeration is necessary to avoid these dead yeast cells.
4) Do not store too long
Beer that contains a lot of yeast and is stored a little too long can contain a lot of dead yeast cells. So take this into account and communicate this to the end consumer.
How do I spot autolysis?
This flavor anomaly logically causes an exaggerated yeast flavor, since we are left with a large number of dead yeast cells.
The taste is sometimes compared to barbecue meat or potato chips and is sometimes described as sulfurous. Depending on the concentration of dead yeast cells in your beer and so you can observe different flavors.
Apart from the taste and aroma, autolysis also affects the head, so there will be less foam on your beer and the head will also be gone faster.
7. GUSHING; overflowing bottle
Gushing or gushing of beer, also called fobbing, is the overflow of beer when you open the bottle. In general, you can say that there are many possible causes here. Namely, anything that promotes an accelerated release of gas can cause gushing.
So chemically, you can say that any particle that forms a “nucleation” site for carbon dioxide causes gas to be released faster.
Thus, one must avoid releasing carbon dioxide too much and too quickly.
“How do I prevent gushing?”
1) Choose quality malt
In almost every case of gushing, defective or infected malt is at the root of the problem. Thus, the most important factor is the quality of the malt.
Defective malt is often a result of harvesting barley under humid conditions. Maltings still often depend on the season, meaning that not every harvest is identical in quality. One possible solution is to mix different malts to reduce the risk.
2) Add calcium salts
If the problem is not with the malt then often calcium oxalate is the culprit.
Oxalate anions, derived from the husks of the grains used in brewing, can precipitate from calcium. Thus, separate crystals are formed. These crystals are the source or germination sites for accelerated CO2 release. This can be a particularly troublesome problem for brewers who produce beers with secondary fermentation in the bottle with high levels of CO2.
Brewers who experience gushing, caused by oxalate crystals, should ensure that sufficient calcium is present at the beginning of the brewing process. This allows the calcium to precipitate from the oxalate BEFORE filtration and bottling.
Unfortunately, this is a sensitive system of chemical balance and addition of calcium is not always successful.
3) Use special hops
Some hops can largely prevent gushing. Thus, an appropriate hop technology can remedy or reduce the problem.
How do I spot gushing?
Does your bottle spontaneously overflow after opening? Did you not shake it? Then you have the problem of gushing. The cause of this problem is certainly not always the brewer’s, if the beer is stored too long, for example, it can also foam later.
Belgian beers often have this problem since our beers tend to be high in carbonation.